Optimized loading effects on pressure steam sterilization of loaned surgical instruments using product family categorization
Study design
This quasi-experimental study employed a non-contemporaneous controlled experimental design. Surgical instruments that were sterilized between January and March 2023 served as the control group, while those sterilized from April to June 2023 constituted the test group.
Study setting and subjects
The research was conducted within the Central Sterile Supply Department (CSSD) of a large tertiary care hospital, which has a capacity of 5,500 beds and an average daily surgical volume ranging from 600 to 800 procedures. The CSSD encompasses a floor area of 3,200 m2 and is equipped with six pressure steam sterilizers (Beliman, MST 9618HS2). Based on a sample size calculation using G*Power software (3.1.9.7 Version), with a medium effect size of 0.3 and a two-sided test with 0.05, the required total sample size was determined to be 172 batches.
(G*Power software URL:https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower)
Ethical considerations
This study exclusively involved loaned surgical instruments and did not include human or animal subjects. All loaned surgical instruments utilized in this project were duly recorded by the hospital. The Institutional Review Board of our institution concluded that the study protocol was exempt from review.
Intervention program
Product family theory
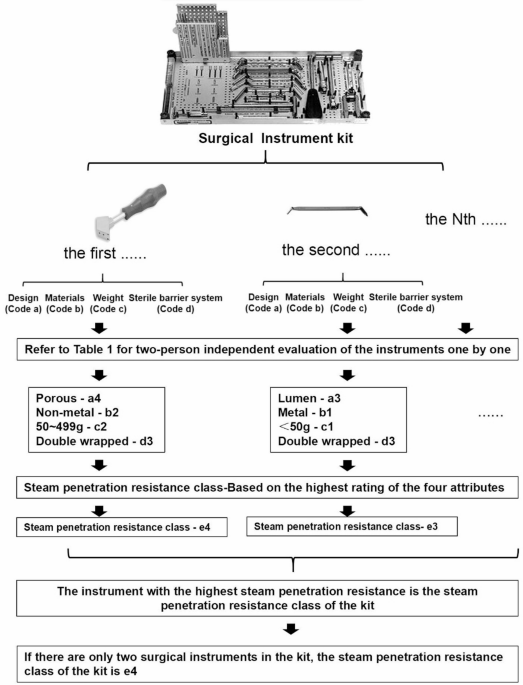
Within the framework of product family theory, the attributes of surgical instruments pertinent to moist heat sterilization (Table 1) are systematically categorized into design (code a), material (code b), weight (code c), sterile barrier system (code d), and steam penetration resistance (code e). Each attribute is further stratified into various levels according to its potential impact on steam penetration resistance. A higher coded number signifies increased resistance to steam penetration. The ultimate class of steam penetration resistance for a kit of surgical instruments is determined by the highest-level attribute within that kit.
Determination of steam penetration resistance class
The research team conducted a meticulous evaluation of each instrument within each kit, adhering to the rating criteria established for general surgical instrument attributes as outlined in the Product Family Categorization Theory (Table 1). An independent assessment by two evaluators was employed to rate the attributes of design, material, weight, sterile barrier, and steam penetration resistance for each loaned surgical instrument. Higher ratings corresponded to increased challenges in achieving effective sterilization. In cases where the two evaluators disagreed on the results, a third evaluator was engaged to assist in the assessment process. After evaluating each kit, the instrument with the highest steam permeation resistance was assigned as the steam permeation class for that kit of loaned surgical instruments (Fig. 1). Initially, the project team conducted an inventory of all loaned surgical instrument kits registered with our hospital, amounting to a total of 98 kits.
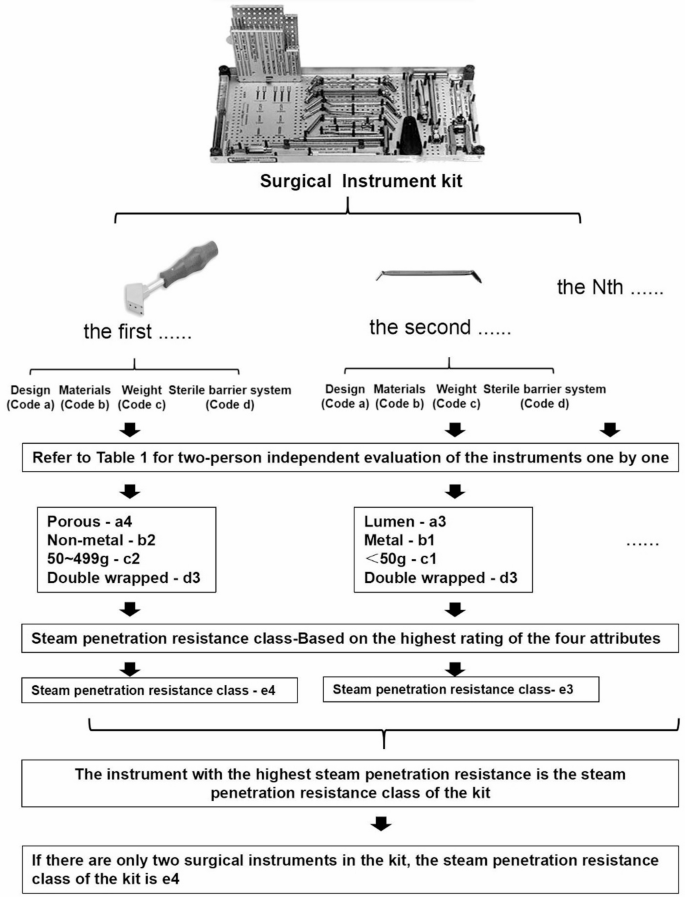
The flow chart the process of evaluating the steam penetration resistance class of surgical instrument kits.
Expert review
The research team convened a review meeting with a panel of 10 experts specializing in CSSD, hospital infection control, and medical devices. The panel comprised 4 senior-level and 6 associate senior-level professionals, all of whom possessed at least a bachelor’s degree. The mean age of the experts was 50 years (SD = 5.53), and their average professional experience spanned 19.5 years (SD = 4.96). During the meeting, the experts evaluated the steam penetration resistance class of various loaned surgical instrument kits using a 5-point importance scale. The authority coefficient for the experts involved in this project was calculated to be 0.854, signifying a high level of expertise in the domains of disinfection, sterilization, and infection control research. The coefficient of variation ranged from 0 to 0.24. The Kendall consistency coefficient was 0.352 (p < 0.001), reflecting a high degree of agreement among experts on the results of steam penetration resistance class of loaned surgical instruments and a credible result of expert opinion consistency.
Determination of sterilization procedures
The pressure steam sterilizer employed in our hospital was equipped with two pre-set sterilization programs for surgical instruments: the standard instrument program and the heavy-duty sterilization program. Based on prior research14, the project team found that surgical instruments made from specialized materials, such as wood, silicone, and high-polymer substances, tend to exhibit temperature delay. This delay could lead to the accumulation of condensation and an increased risk of wet packs. According to the theory of product series, the steam permeation resistance of instruments manufactured from these specialized materials typically ranges from e5 to e6. Consequently, the drying time required for these instruments was longer than that of the standard instrument procedure, aligning with the heavy-duty sterilization procedure in this sterilizer. The relationship between specific sterilization procedure parameters and steam permeation resistance is detailed in Table 2.
Implementation
The team systematically recorded the steam permeation resistance classes of all loaned surgical instruments in a computerized format within the existing information traceability system. This was done to facilitate the ease of operation for the staff. Prior to the project’s implementation, all staff members received training to perform system maintenance. Consequently, personnel responsible for packaging could distribute the loaned surgical instruments based on their steam permeability resistance classes, as indicated on the traceability labels. Instruments were then placed in different loading racks according to their resistance classes to optimize the sterilization process. Following a unified loading process, the sterilization staff scanned the traceability bar code of each instrument kit individually. For the test group, the sterilization procedure was selected based on the highest steam penetration resistance class within the batch. In contrast, the control group based their selection on the oversized and overweight instrument markings on the items to be sterilized. Specifically, the heavy-duty instrument procedure was employed when oversized and overweight instruments were included in the batch, whereas the standard instrument procedure was utilized for standard weight. All loaned surgical instruments underwent the sterilization cycle according to the designated sterilization program. To minimize bias arising from variations in sterilizers, only pressure steam sterilizers No. 6 or 5 were employed for the project implementation period.
Outcome measurement
Selection of sterilization procedure
The selection of sterilization procedure was observed in two groups, both prior to and following the implementation of the optimized loading protocol. The procedures included both standard instrument procedure and heavy-duty instrument procedure.
Sterilization monitoring result
Sterilization monitoring encompasses physical, chemical, and biological assessments, as well as wet pack inspections. Physical monitoring is defined by maintaining a temperature fluctuation range within ± 3℃ during sterilization, a sterilization duration of no more than 5 min, and a sterilization pressure between 201.7 kPa and 229.3 kPa. Both chemical and biological monitoring employ the 3 M Attest™ Super Rapid 5 Steam-Plus Challenge Pack (41482 V), which includes an integrated Type 5 Chemical Integrator and a Super Rapid Readout Biological Indicator. Successful sterilization is indicated when the black reagent on the Type 5 Chemical Integrator advances to the “ACCEPT” window. The Super Rapid Readout Biological Indicator is incubated in a 3 M Rapid BioReader at 56℃ for 24 min, a negative result signifies a successful sterilization process. For wet pack inspection, after a cooling period of 30 min, both the interior and exterior of the instrument package, as reported by the operating room nurse or CSSD staff, must be free of visible water droplets or moisture. All these criteria must be simultaneously met to confirm successful sterilization.
Data collection
Following the completion of the sterilization cycle, an independent quality inspector, who is not involved in the project, systematically documents the procedures, as well as the results of physical, chemical, and biological monitoring, and assesses the wet pack conditions for each batch of sterilizers.
Statistical analysis
Statistical analyses were conducted using SPSS version 22.0 software. Categorical data were analyzed using the chi-square test. The significance level was set at α = 0.05, with differences considered statistically significant at P < 0.05.
link